Chances are 9 in 10 that when you reach into your medicine cabinet or seek treatment at the clinic, you’re encountering the power of curiosity-driven research. This is research defined by the pursuit of basic questions rather than immediate products and profits. How does this work? Why does this happen?
Answers to these fundamental questions build towards incredibly useful, even life-saving destinations. But that path proves long and unpredictable, with new branches frequently shooting off into unforeseen directions. And seeming dead-ends abound: “failures” (we learned what doesn’t work) often outnumber successes (it all went to plan).
Curiosity-driven research is unique, and it’s under threat. That’s why we’re bringing its defining features into sharper focus and amplifying the voices in our community who know this world inside and out.
The long game
It’s easy to associate success with speed in a world where we can, in a couple of clicks, take a video call from halfway across the planet while selecting same-day shipping on a new gadget. (Both technologies stem from curiosity driven-research.)
Curiosity-driven research doesn’t promise quick results or profits. Though few of us see this work as it unfolds, it lays the foundation for discoveries that save millions of lives.
More than 30 million Americans take statins to reduce their risk of cardiovascular disease. One in 8 have taken a GLP-1 drug. 40 million MRIs are performed in the U.S. each year, helping to diagnose everything from brain tumors to torn knee joints. And COVID-19 vaccines are estimated to have prevented over 14 million deaths. Behind these enormous numbers are individuals doing their best to navigate health, sending ripple effects across even greater numbers of people — families, workplaces, communities.
And behind these impacts are generations of scientists building on discoveries unearthed by curiosity-driven research over decades, even centuries.
 The discovery of cholesterol, for example, dates to 1815, just over 170 years before the approval of lovastatin as a treatment to reduce the risk of coronary artery disease. In between is a plodding timeline punctuated by a few early descriptions of arterial plaque buildup and incremental insights into the basic science of cholesterol synthesis and inhibition. In the 1970s, scientists first in Japan and then in the U.S. isolated statins from fungi. Nearly another decade passed before the FDA approved of lovastatin as a drug.
The discovery of cholesterol, for example, dates to 1815, just over 170 years before the approval of lovastatin as a treatment to reduce the risk of coronary artery disease. In between is a plodding timeline punctuated by a few early descriptions of arterial plaque buildup and incremental insights into the basic science of cholesterol synthesis and inhibition. In the 1970s, scientists first in Japan and then in the U.S. isolated statins from fungi. Nearly another decade passed before the FDA approved of lovastatin as a drug.
Researchers at Novartis Institutes for BioMedical Research and Harvard University performed this kind of analysis for over 30 drugs and found that the median time from scientific discovery to an approved therapy is more than three decades.
Science is about what we don’t know. The most exciting science is about forging new directions and discovering things that haven’t been imagined before.

Paul Ahlquist
Investigator, Virology
By contrast, the accelerated effort to bring COVID-19 vaccines to market might seem lightning fast. Only 16 months separate the first reported COVID-19 cases and wide availability of vaccines in the U.S. But decades of foundational, curiosity-driven research had laid the groundwork for this unprecedented effort.
That work started with a handful of research groups making their way towards the discovery of messenger RNA in the mid-twentieth century. Indeed, an earlier and ongoing motivation behind mRNA vaccines focuses on fighting cancer.
 In 1984, researchers engineered biologically active mRNA in the laboratory for the first time, with the idea of using it to study how genes work. A few years later came the demonstration that human cells could absorb mRNA encapsulated in lipid droplets and produce proteins. And in 2005, scientists discovered how to modify synthetic mRNA to prevent the immune system from degrading it before it could deliver its message: the RNA transcript that could teach the cell how to fight off a viral attack.
In 1984, researchers engineered biologically active mRNA in the laboratory for the first time, with the idea of using it to study how genes work. A few years later came the demonstration that human cells could absorb mRNA encapsulated in lipid droplets and produce proteins. And in 2005, scientists discovered how to modify synthetic mRNA to prevent the immune system from degrading it before it could deliver its message: the RNA transcript that could teach the cell how to fight off a viral attack.
Each of these milestones represents a critical step in the chain of discoveries that ultimately made rapid mRNA vaccine deployment possible.
Centuries-old science gave us the first observations of what would become entire fields like cell biology and quantum physics. Today we live in an era of use-inspired accelerated discovery. But this pattern, where findings build over many years towards sometimes unforeseeable transformative innovations, remains a core feature of basic science. And that also means the scientists who do this work choose to pursue long-term visions, knowing their impacts may be felt only decades later.
Prepared minds favored by chance
Pursuing curiosity also means the path to innovation can be unpredictable, with scientific advances emerging from seemingly unrelated work, sometimes across entirely different fields.
In the summer of 1961, two Princeton scientists were brought to Friday Harbor, off the Washington coastline, with a simple question about the jellyfish Aequorea victoria: why does it glow? In their studies analyzing the “squeezate” they collected from thousands of jellyfish, they discovered what later became known as green fluorescent protein, or GFP.

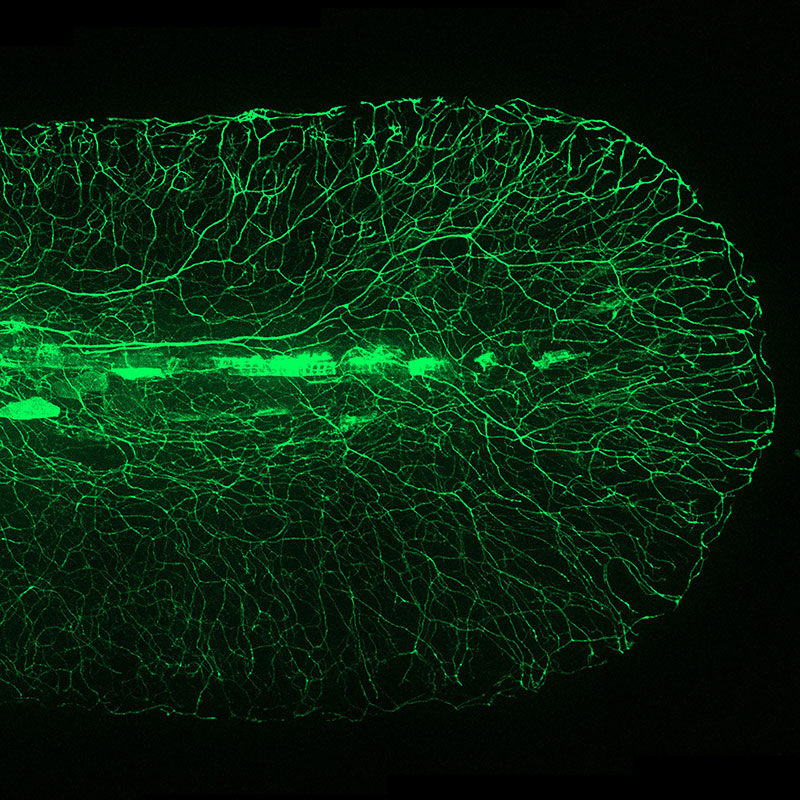
GFP lighting up the neural network in the tail of a live zebrafish Credit: Pui-Ying Lam/Kevin Eliceiri
A quarter century later, Martin Chalfie was sitting in a seminar when he learned about GFP from a visiting speaker. As a researcher focused on the transparent model organism, the worm Caenorhabditis elegans, Chalfie immediately realized the exciting potential of GFP to visualize the worm’s nervous system — and set about work to make this possible.
It’s a moment that forever changed biomedical research. Today GFP and other fluorescent proteins are routinely used to visualize subcellular life with a precision that had been unimaginable without them. Introducing these proteins into cell lines or model organisms to “tag” particular molecules of interest allows us to track processes like the progression of disease, the activity of drugs against their targets, gene expression, and more.
Basic science allows scientists to ask questions about how the world works. And when you do that, you don’t know what you’re going to find. Most of the major breakthroughs in our understanding of human health came from looking at unexpected places.

Phil Newmark
Investigator, Regenerative Biology
From fundamental questions, surprising and life-changing outcomes

Eight years of Nobel-prize winning work, building on discoveries in the ancient immune systems of bacteria and Archaea led to…

“Genetic scissors,” or CRISPR-Cas9, a ubiquitous tool used across the life sciences to alter virtually any organism’s DNA — helping us unlock the code of life and bringing the dream of curing inheritable diseases within reach.

More than a century of work on gastrointestinal hormones, and more than a decade of work building on discovery of a more stable analog found in Gila monster venom led to…

A new class of diabetes and obesity drugs known as “GLP-1s,” now also under research for treating addiction disorders and neurodegenerative diseases

One scientist’s decade-long belief, against the Central Dogma, that genetic information can flow not just from DNA to RNA but in the reverse — plus two experiments published alongside each other that independently proved it led to…

The 1970 discovery of reverse transcriptase, a paradigm shift with impacts across science and medicine — including the rapid identification of HIV just over a decade later, along with antiviral therapies to treat it; new biotechnologies, such as the PCR tests made widely recognizable during the COVID-19 pandemic; and new inroads into the study of cancer biology and treatments.

Physics experiments giving the first evidence of nuclear magnetic resonance in the 1930s and 40s and laying the foundation for a next generation of chemists and physician scientists to create magnetic resonance imaging (MRI) technology in the 1970s led to…

Atomic clocks — without which we would not have accurate GPS navigation or secure financial transactions in global trading markets — and in the world of medicine, a revolution in non-invasive diagnostic imaging: MRI is used on nearly every organ system, including powerfully in the nervous system and brain
Science is full of such stories, from CRISPR to GLP-1s to GPS, teaching us to expect the unexpected.
Among them is the Wisconsin story behind the blood thinner warfarin. In February 1933, Ed Carlson, a dairy farmer from Saint Croix County, braved a blizzard to bring his dead cow to UW–Madison, seeking answers about why she and many others had recently fallen victim to internal bleeding. A Saturday afternoon, Carlson had been looking for the state veterinarian, but wound up at the lab of Karl Paul Link with his cow, a churn full of blood, and 100 pounds of hay the cow had been eating.
Like Martin Chalfie, Link was in the right place at the right time. In Link’s telling, the visit was a “direct catalytic hit” from agriculture to spur lab research into what he immediately recognized as sweet clover disease. First, Link and his colleague, who was particularly mesmerized by the lack of clotting capacity in the blood that he rubbed through his fingers, sent the farmer home frustrated that it was with “promises [that] might come true in five, ten, fifteen years, maybe never.”

Karl Paul Link, professor of biochemistry and discoverer of warfarin, performs a laboratory procedure with fellow researcher Mark A. Stahmann. Photo: Gary Schulz. From UW–Madison Collections
Then the lab spent the next seven years trying to isolate the anticoagulant agent, dicumarol, and another eight finding a more appropriate derivative for clinical use. This led to the patenting of the rodenticide warfarin (named for the first-of-its-kind patenting agency WARF, or the Wisconsin Alumni Research Foundation). Finally, after yet more work to refine dosing for human use, naturally occurring cow poison became not just patented rat poison but a lifesaving drug for humans, Coumadin, preventing potentially fatal blood clots.
However whimsically fated such stories seem, they also represent the payoffs of allowing researchers the freedom to crack away at their curiosity, day after day. That freedom is as integral to modern science as in eras past — but arguably harder to come by.
Productive partnerships
Since World War II, the federal government has been one of the biggest funders of curiosity-driven science at research institutions across the country. This investment has produced countless health advances, kept the world safe from once-common diseases, and has trained the scientific workforce that can respond to emerging threats.
It has also generated profound economic growth. In Wisconsin alone, funding from the National Institutes of Health supported more than 6,500 jobs and $1.38 billion in economic activity in 2024, according to United for Medical Research. Nationally, that number is over 94 billion, translating to $2.56 of economic activity for every $1 of research funding.
The most successful companies are the ones who go back to the basic science, and, and don’t just develop the product. They really, truly start with that curiosity-driven insight and, and keep a team of people who are focused. They are looking they’re scouring the universe, the world for the best ideas and and at some point, the alchemy of what they do kind of becomes real. And they convince a product development person to take that idea and fuse it into a product.

Kevin Conroy
CEO, Exact Sciences
 But it’s not just federal support that makes this work possible. The total share of basic research footed by federal funders has in fact decreased relative to increases from the business and philanthropy sectors over the last several decades, going from 70% of total funding for basic science in the 1960s down to 51% in recent years.
But it’s not just federal support that makes this work possible. The total share of basic research footed by federal funders has in fact decreased relative to increases from the business and philanthropy sectors over the last several decades, going from 70% of total funding for basic science in the 1960s down to 51% in recent years.
Even before proposed cuts to federal funding threw the world of basic research into damaging uncertainty in 2024, many have been concerned about funding trends, including that decline in the federal share and a tendency to favor incremental pre-proven advances. Private philanthropy plays a particularly key role in supporting basic science that ventures into greater unknowns or takes on a greater level of risk.
Curiosity lays the essential foundation for successful applied research and translation to products and profits. Shaking the foundation of broad, blended funding for basic science compromises the integrity of this entire ecosystem and the impacts we can expect to emerge from it.
Fearless Science at Morgridge
Following where curiosity leads in in this landscape is often an act of courage.
The Morgridge Institute takes a distinctive approach to curiosity-driven research: Fearless Science. This is our commitment to providing the freedom and flexibility that scientists need to take risks that will advance new knowledge, and this ethos is rooted in our origin story. Jamie Thomson’s basic research into embryo development in non-human primates led to his pioneering work in stem cells, a path that would lead in turn the founding of the Morgridge Institute in 2006.
One analogy would be that Morgridge can be like the stem cells of the research world. We have the flexibility and potential to become almost anything.

Jing Fan
Investigator, Metabolism
Now we are home to 20 investigators who continue to pursue new frontiers in biology — alongside new groundbreaking technologies and new understandings of the deep connections between science and society.
Some of the questions we’re following take us:
At Morgridge, I’m able to pursue high-risk projects and I’m given the time to do what I feel is important. I tell people Morgridge is like a utopia for research.

Melissa Skala
Investigator, Biomedical Imaging
Why it matters
 Without sustained support for curiosity-driven research, the engine behind life-saving discoveries will falter and talented researchers may leave the field. Investing in curiosity-driven research builds the foundation for the breakthroughs of tomorrow.
Without sustained support for curiosity-driven research, the engine behind life-saving discoveries will falter and talented researchers may leave the field. Investing in curiosity-driven research builds the foundation for the breakthroughs of tomorrow.
On the most fundamental of levels, this support speaks to a human need to know more about why we’re here and where we’re going.
Our only limit is our curiosity.
Humans, it turns out, are innate scientists. They’re curious. They want to explore, to understand things. Kids drive their parents crazy by asking how and why every five minutes, they take apart their toys and your gardening tools to understand how they work. They’re thrilled by discovery. […] So basic scientists are humans who choose to engage that curiosity, that innately human characteristic, for a living — to know things that no one else knows.

Keith Yamamoto
Morgridge Trustee